Biological Metamaterials for Influencing Cell Behaviour
We use nanostructured surfaces to study cell response to different microenvironments, including the use of high-aspect-ratio nanostructures. These can be used to study mechanotransduction, as well as provide topographical guidance.
Our methods
We explore how varying design parameters such as the pitch and sharpness influences the response of different cell types. The aim is to both understand how the cell--material microenvironment influences behaviour, and to explore the structure-property relationships of biomaterials.
We use microfabrication technologies, more commonly used to make electronic devices, to nanostructure the surface of different materials with well-defined patterns. We work with collaborators to culture cells on these surfaces and then use high-throughput image analysis to understand cell responses.1
Impact of high-aspect-ratio nanostructured surfaces on cells
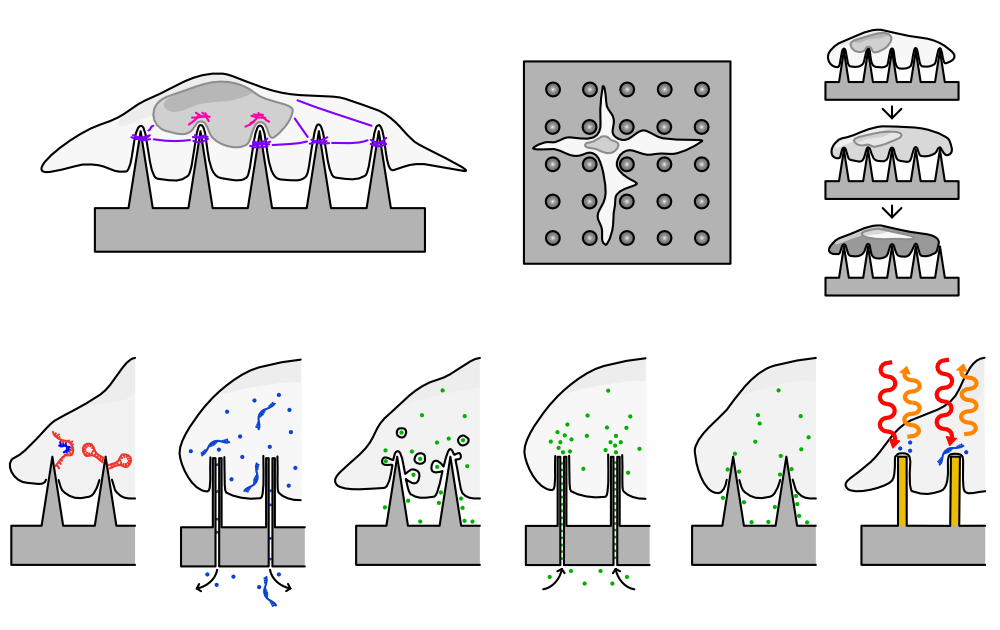
Cells typically change their shape and behaviour on nanostructured surfaces.2 Some can form highly elongated shapes, aligning to the underlying structures - a behaviour which can be useful in tissue engineering, where the aim is to recreate the patterns of cells found within tissues.
Some cells contort around the sharp tips of the structures. These dramatic shape changes can be useful in drug-delivery, where the nanostructures can be coated with a particular molecule or drug, which is then brought into very close contact with the cell.
The relationship between the size or shape of a nanostructure and quantifiable changes in a group of cells is less well understood. Do all cells behave the same? Does a single cell alone respond the same as a cell with many neighbours? This is where my research aims to provide answers. For example, in a recent study led by Dr Hyejeong Seong, we found that sharpness of the nanostructures had a direct effect on the shape and behaviour of human mesenchymal stem cells, an insight that influences how we design biomaterials moving forward.3
Read our review on the applications and techniques for high-aspect-ratio nanostructures
Applications to prokaryotic cells
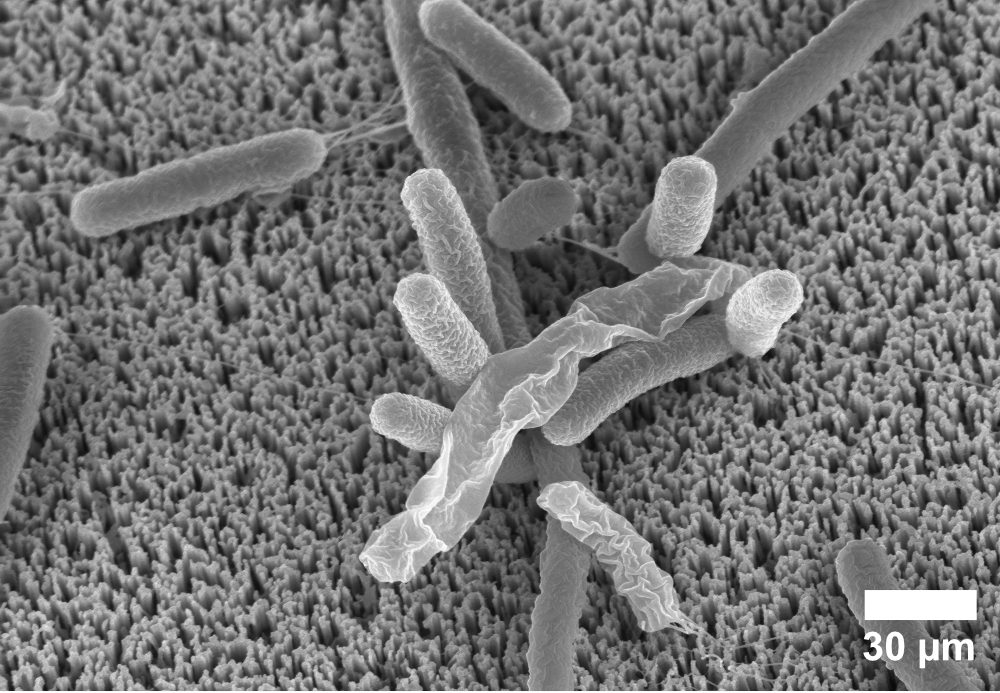
Nanostructuring is also being explored to create antibacterial surfaces, physically disrupting the growth of bacteria, rather than using a chemical agent. This is inspired by nature - some species of cicadas have nanostructured wings that are thought to act as antibacterial surfaces.
Stuart worked with Dr Michele Becce and Dr Anna Klöckner to explore how nanowires (an even tinier, denser version of nanoneedles) could be applied to bacteria such as E. coli. Fascinatingly, they found that in many cases, bacteria were able to survive on our nanostructures.
This important negative result shows that simply adding nanostructures is not enough - that antibacterial behaviour most likely relies upon a combination of factors. We were also able to make several key recommendations about how to improve the assessment of anti-bacterial viability on nanostructured surfaces.4
Read our key recommendations for improving bacterial-nanostructure interfacing experiment
Understanding fundamental behaviours of nanoscale structure
Computer modelling allows us to overcome the limitations of experiments by simulating nano-environments smaller than our current experimental tools allow us to probe.
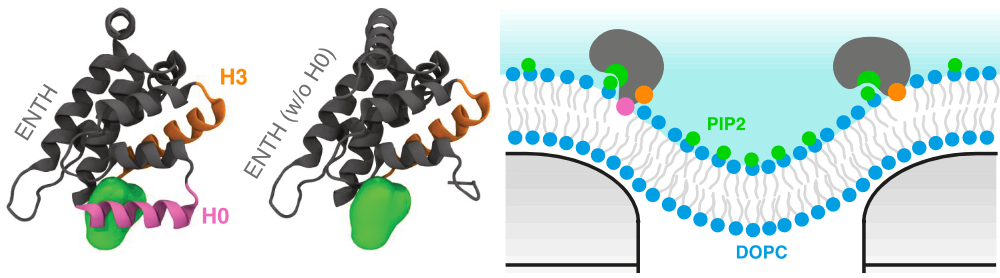
Stuart worked with Dr Alexis Belessiotis-Richards to explore the effects of nanoscale curvature on cell membranes. This work revealed how tiny distortions to a cell membrane can cause parts of proteins to bind to these regions - an important precursor to how materials are transported across the cell membrane.5
Read about the impact of nanoscale curvature on protein localisation
Parts of this research are an ongoing collaboration with The Sero Lab at the University of Bath, and The Stevens Group at Imperial College London/University of Oxford.
-
Reprinted/adapted by permission from: Higgins SG, Becce M, Seong H, Stevens MM, Nanoneedles and Nanostructured Surfaces for Studying Cell Interfacing, 2020, 7th International Conference on the Development of Biomedical Engineering in Vietnam (BME7) by Vo Van Toi, Trung Quoc Le, Hoan Thanh Ngo, Thi-Hiep Nguyen © 2020. DOI: 10.1007/978-981-13-5859-3_37 ↩
-
Redrawn from: Higgins SG, Becce M, Belessiotis-Richards A, Seong H, Sero JE, Stevens MM, High-Aspect-Ratio Nanostructured Surfaces as Biological Metamaterials, 2020, Advanced Materials. DOI: 10.1002/adma.201903862 ↩
-
Seong H, Higgins SG, Penders J, Armstrong JPK, Crowder SW, Moore AC, Sero JE, Becce M, Stevens MM, Size-Tunable Nanoneedle Arrays for Influencing Stem Cell Morphology, Gene Expression, and Nuclear Membrane Curvature, 2020, ACS Nano. DOI: 10.1021/acsnano.9b08689 ↩
-
Reproduced under terms of CC-BY license from: Becce M, Klöckner A, Higgins SG, Penders J, Hachim D, Bashor CJ, Edwards AM, Stevens MM, Assessing the Impact of Silicon Nanowires on Bacterial Transformation and Viability of Escherichia Coli, 2021, Journal of Materials Chemistry B. DOI: 10.1039/D0TB02762F ↩
-
Adapted under terms of CC-BY license from: Belessiotis-Richards A, Higgins SG, Sansom MP, Alexander-Katz A, Stevens MM, Coarse-Grained Simulations Suggest the Epsin N-Terminal Homology Domain Can Sense Membrane Curvature without Its Terminal Amphipathic Helix, 2020, ACS Nano. DOI: 10.1021/acsnano.0c05960 ↩